Approach
A central premise of my research is that glycemic harm may depend not only on glucose concentration, but on a dynamic metabolic state: the direction and speed of glucose changes, the accompanying insulin activity, and the recent history of glucose exposure.
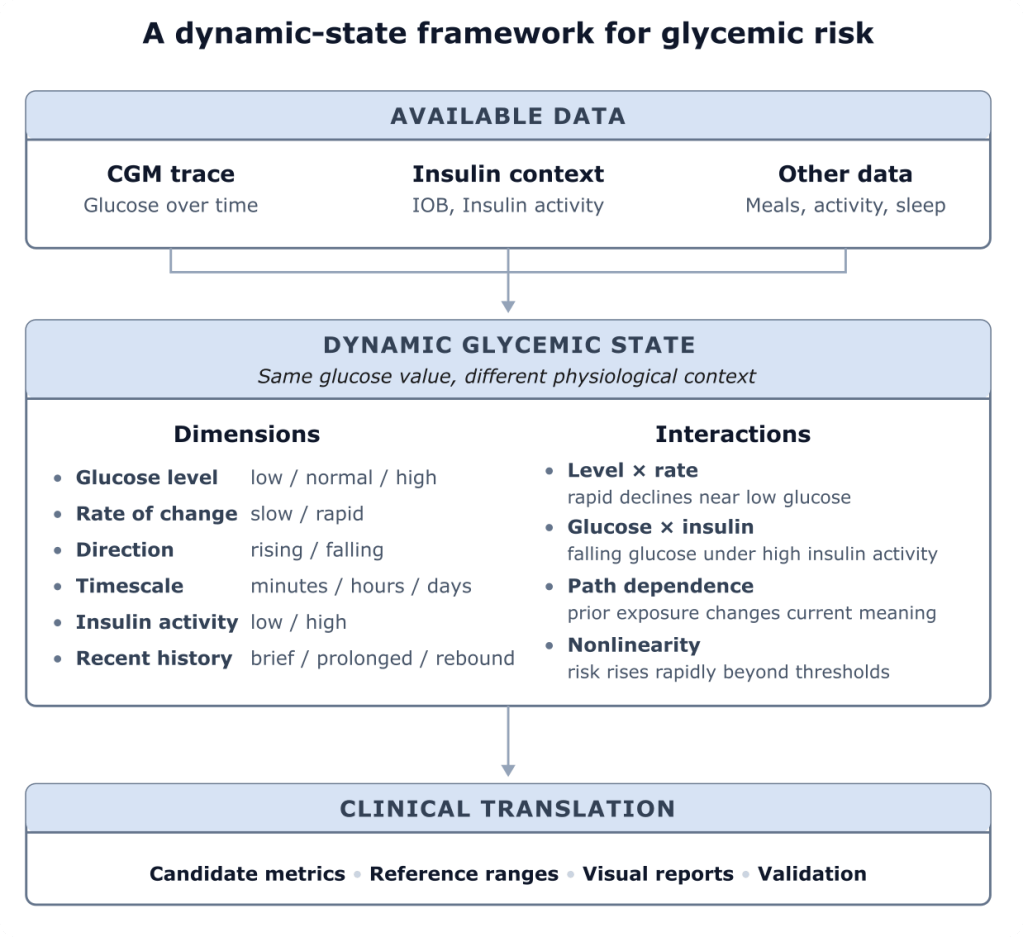
Hypotheses
I am particularly interested in evaluating the following hypotheses:
- Rate of change: faster glucose transitions may be more physiologically stressful than gradual changes.
- Direction: rapid declines may be especially relevant because of autonomic activation and counterregulatory stress.
- Timescale: short-timescale changes, particularly over 5–15 minutes, may carry clinically important information.
- Nonlinearity: risk may rise disproportionately beyond physiological rate-of-change thresholds, while changes slower than these thresholds, even if frequent, may be relatively benign.
- Glucose level–rate interaction: rapid declines in the lower glucose range, including above the formal hypoglycemia threshold, may be more harmful than equivalent declines at higher glucose levels.
- Glucose–insulin interaction: hypoglycemia or rapid declines under high insulin activity may represent a different physiological state, not merely a different glucose-risk state, than glucose data alone indicate.
- Path dependence: the recent history of the trace may change the meaning of the current glucose state; for example, a rapid fall after prolonged hyperglycemia may not be equivalent to a brief excursion that quickly returns to baseline.
These are working hypotheses, in part derived from clinical observations, intended to be tested, refined, or rejected through further analysis.
Current research directions
Glucose variability as dispersion and volatility
Reframe glycemic variability as two complementary dimensions: glucose dispersion (tight versus dispersed) and glucose volatility (stable versus volatile), and identify physiological reference ranges for normal/abnormal volatility.
Hypoglycemia phenotyping
Develop richer descriptions of hypoglycemia that include not only glucose depth and duration, but also onset speed, insulin activity, recovery pattern, and recent glucose history.
Glucose–insulin interaction
More generally, use combined CGM and insulin data to identify metabolic states that may be underestimated by glucose-only metrics, especially during hypoglycemia or rapid declines under high insulin activity.
Visualisation and reporting
Develop visual summaries, such as the ambulatory volatility profile, that make glycemic dynamics interpretable within familiar CGM-report formats.
Validation
Because diabetes outcome data are sparse and confounding is substantial, the aim is not purely black-box prediction from large datasets. The approach is pattern-first: identify physiologically interpretable signals in individual glucose–insulin traces, formulate hypotheses from first principles, and then test whether they generalise in larger datasets.
Validation therefore needs to proceed across a spectrum of evidence, from mechanistic reasoning, physiological reference ranges, illustrative cases, and trace-level analysis, through to retrospective cohorts, symptom associations, complication outcomes, and eventually prospective or interventional studies.